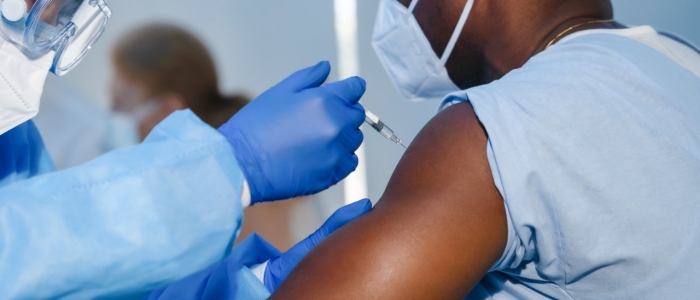
With concerns still lingering about the safety and side effects, the Pfizer/BioNTech COVID-19 vaccine has passed the full approval process and it is ready for distribution in the U.S. The first doses will arrive at California hospitals this week.
On Dec. 11, the U.S. Food and Drug Administration (FDA) issued an emergency use authorization for the Pfizer/BioNTech coronavirus vaccine to be distributed to individuals 16 and older. In a statement, the FDA said that Pfizer’s clinical trial data showed that the “known and potential” benefits of the vaccine outweighed its risks.
“Today’s action follows an open and transparent review process that included input from independent scientific and public health experts and a thorough evaluation by the agency’s career scientists to ensure this vaccine met FDA’s rigorous, scientific standards for safety, effectiveness, and manufacturing quality needed to support emergency use authorization,” said FDA Commissioner Stephen M. Hahn, M.D.
On Dec. 12, U.S. Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices recommended the Pfizer vaccine. CDC Director Robert Redfield, M.D., accepted the recommendation on Dec. 13, passing the vaccine through the final step needed for distribution. On the same day it received CDC. approval, the first batch of vaccines was shipped out of a Pfizer plant in Portage, Michigan.
“This is the next step in our efforts to protect Americans, reduce the impact of the COVID-19 pandemic, and help restore some normalcy to our lives and our country,” Dr. Redfield said in a statement.
In addition to governmental reviews, state officials and public health organizations are also reviewing the safety of the Pfizer vaccine. The Western States Scientific Safety Review Workgroup, made up of scientists and experts representing California and other states, independently reviewed and endorsed the vaccine’s safety on Dec. 13.
“This morning, the Workgroup recommended the Pfizer vaccine as safe for public use. With shipments of the vaccine soon on their way to California, we are working hand-in-hand with local public health officials to get the vaccine out to the first phase of recipients. Their work will continue as data becomes available on other potential vaccines,” said Governor Gavin Newsom.
The National Medical Association, the largest national organization representing African American physicians and patients, has also formed a COVID-19 task force to offer a recommendation regarding the vaccine.
California state officials expect to receive 327,000 doses of the Pfizer vaccine in the upcoming week. The Moderna coronavirus vaccine is also expected to be approved by the FDA this month, and it’s expected that California will receive about 2.16 million doses of the Pfizer and Moderna vaccines combined by the end of December.
The Pfizer vaccine is given as two shots, administered 21 days apart. FDA documents note that the vaccine seems to provide “some protection” after the first shot, but it takes both doses to reach an efficacy of 95 %.
Once the vaccines arrive in California, they will be sent to hospitals and other facilities that can store them at around minus 94 degrees Fahrenheit. Storage facilities confirmed so far include Cedars-Sinai Medical Center and UC San Francisco Medical Center.
Priority for receiving the vaccine will go to health workers most likely to be exposed to COVID-19, as well as residents and staff of nursing homes and long-term care facilities. According to public health experts, vaccines will most likely be available for the general public in spring or summer 2021.
As of Sunday night, the COVID-19 death toll in the United States since the pandemic was declared in March reached nearly 300,000. An estimated 16.2 million Americans have been infected. In California, there have been an estimated 21,043 deaths and 1.5 million reported cases.
According to FDA documents, the vaccine does have some common adverse effects, including pain at the injection site, fatigue, headache, chills, muscle or joint pain and fever. Long term effects of the vaccine are not yet known.
The FDA has also warned against using the Pfizer vaccine for people who have had severe allergic reactions to ingredients used in this vaccine. Though severe allergic reactions to vaccines are rare, some participants in the Pfizer trial did experience adverse reactions, and the trial itself excluded people with a history of the severe allergic reaction anaphylaxis.
In a Dec. 12 press conference, the FDA said it will continue to monitor severe allergic reactions associated with the vaccine.